 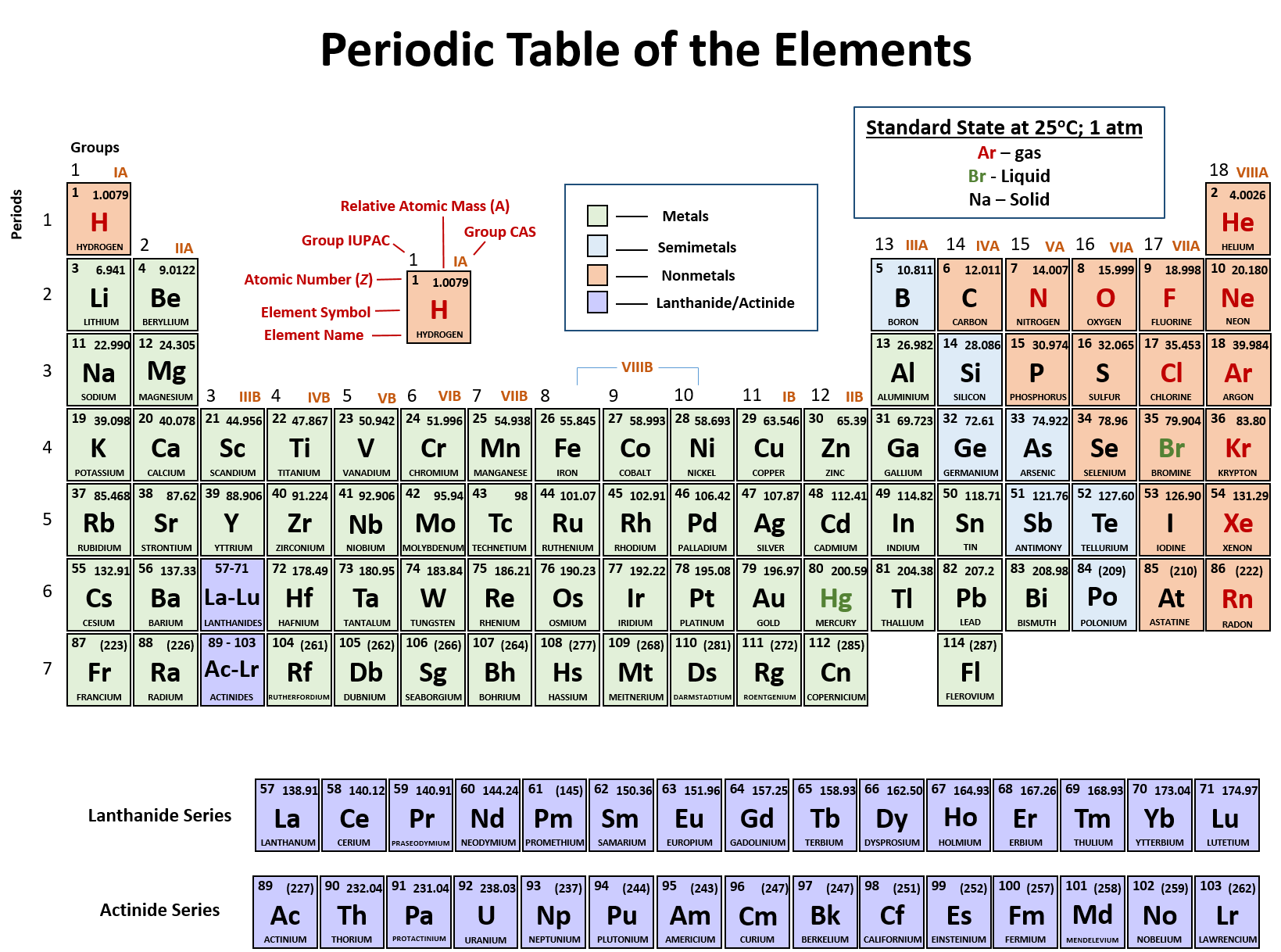
The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. The neutron is slightly heavier than the proton.There are two reasons for the difference between mass number and isotopic mass, known as the mass defect: For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Note that each element may contain more isotopes. How does the atomic number determine the chemical behavior of atoms? Atomic Mass of Krypton Since the number of electrons is responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. 1995 - 2023.Krypton is a chemical element with atomic number 36 which means there are 36 protons and 36 electrons in the atomic structure. Periodic Table of Elements - Krypton - Kr. If you need to cite this page, you can copy this text: This database focuses on the most common chemical compounds used in the home and industry. Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. Molar Mass Calculations and Javascript Calculator.Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Krypton MenuĪ list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page. Additional Notes: Although krypton is a harmless gas, it could asphyxiate if it excluded oxygen from the lungs.Uses of Krypton: Used in fluorescent bulbs, flashbulbs, UV lasers and as a wavelength standard.Around 8 tons of krypton is produced each year world wide. Sources of Krypton: Forms 1 millionth of the atmosphere.Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Specific Heat: 0.248J/gK Regulatory / Health.Description: Colorless, odorless, tasteless noble gas.Coefficient of lineal thermal expansion/K -1: N/A.Valence Electron Potential (-eV): Physical Properties of Krypton.Electronegativity: N/A (Pauling) 2.94 (Allrod Rochow).Valence Electrons: 4s 2p 6 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 48.Number of Electrons (with no charge): 36.Electrons per Energy Level: 2,8,18,8 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6d 10 4s 2p 6.Crystal Structure: Cubic face centered.Cross Section (Thermal Neutron Capture) σ a/ barns : 25. 
Swedish: Krypton Atomic Structure of Krypton.Series: Noble Gasses Krypton's Name in Other Languages.Potential Parent Nuclides Overview of Krypton. 
In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
